-
For a FREE
Consultation -
Current wait:
10 seconds
*By clicking the Submit button, I consent to being contacted by Alexander Shunnarah Personal Injury Attorneys at the number I provided. I also consent to receiving advertisements and telemarketing messages by text message or pre-recorded call, either of which may be dialed by an autodialer. I acknowledge that my consent is not required for purchase, that standard message and data rates apply, and that clicking Submit constitutes my electronic signature for my consent to being contacted and my agreement to the Terms and Conditions.
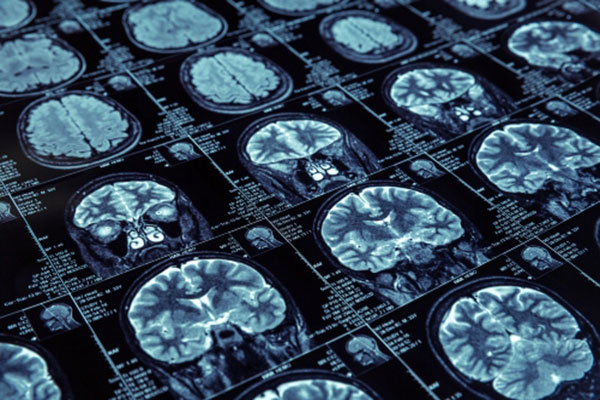
Gadolinium MRI contrast is a dye injected into patients before MRIs to contrast normal and abnormal tissue. This product is injected into a patient’s vein where it gathers in abnormal tissue. Then, when an MRI is conducted, the abnormal areas show up differentiated from normal tissue. After injected, the kidneys quickly clear gadolinium from the body. Although iodine based contrast agents have been used for many years, they have been linked to acute renal failure. In 1999, gadolinium contrast agents were approved for use in MRI. When used in patients with renal failure, it has been reported to cause nephrogenic systemic fibrosis/nephrogenic fibrosing dermopathy (NSF/NFD), which is a serious condition characterized by fibrosis of the skin, joints, eyes, and internal organs. Over time the joints of a person with NSF/NFD will harden, limiting their mobility. This can lead to patients being bound to a wheelchair and even to death.
NFS is an extremely rare fibrosing disorder that is associated with skin changes and scarring of organs in renal failure patients. Symptoms of NFS include changes in the appearance of the skin that are very similar to the changes brought about by scleroderma. Symptoms include redness of the skin, burning or itching sensations, and swelling of the skin. The patients may find skin stretched tight and rigid and have difficulty bending their knees and elbows. There may be muscular weakness and pain. Autopsies of NFS patients have revealed scarring of the internal organs.
There are five gadolinium based contrast agents currently approved for use in the United States:
Because of incomplete data, it is not possible to know if the extent of risk for developing NSF is the same for all agents. Therefore, patients should be screened for kidney problems prior to undergoing MRI with one of these imaging agents. Although NSF has been identified following both single and multiple administrations of gadolinium based agents, it has specifically been identified following sequential administration of the following:
In a healthy individual, gadolinium is removed from the body by the kidneys. When the kidney functioning is compromised, the gadolinium remains and causes serious harm. NSF/NFD can take weeks or months to appear in patients, but once it begins the disease develops rapidly and can be fatal. If you have undergone a MRI or MRA and are experiencing any symptoms which related to NSF/NFD, you should seek immediate medical attention.
Symptoms of NSF / NFD include the following:
Alexander Shunnarah Trial Attorneys is known nationwide for providing outstanding legal advice and excellent representation. We have recovered over $1 billion in lawsuits involving 18 wheeler trucking litigation, wrongful death litigation, commercial vehicle litigation, motorcycle litigation, catastrophic litigation, and mass torts involving medical devices and bad drug litigation. Our prowess in the legal arena comes from our combined years of trial and complex litigation experience.
We founded our practice on the belief that the 7th amendment and the civil jury system is the best means to provide compensation to injured parties and deter wrongdoers from injuring others in the future.














Learn More Through
Our Social Media
Call mE
Alabama Athens Atlanta Bedford Birmingham Boston Buffalo Dallas Gulfport Georgia Houston Huntsville Little Rock Memphis Milwaukee Mobile Montgomery Nashville New Orleans Panama Pensacola Prattville Selma Tallahassee Indiana Washington DC